Setra SRPM Room Pressure Monitor
Designed for critical low differential pressure applications that require stringent pressure monitoring and alarming.
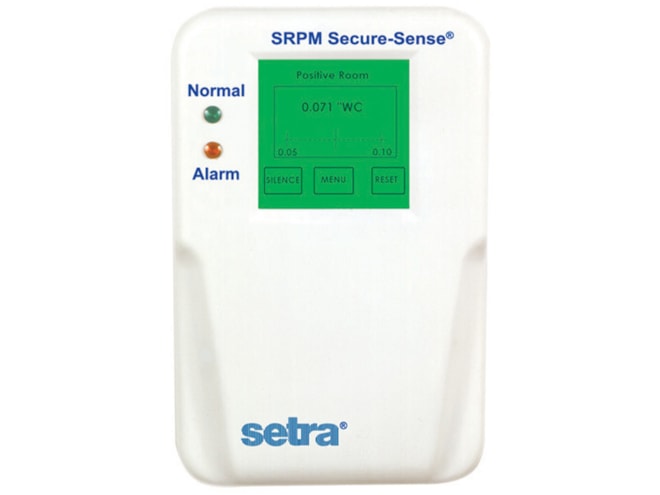
Overview
Features
- Easy, menu-driven programming
- Large LCD touch screen display with bar graph
- Up to ±0.25% accuracy (±0.5% standard)
- Visual and audible local alarms w/ alarm silence
- Optional remote annunciator available
- Selectable analog voltage and milliamp outputs
- SPDT alarm relay
- Red and green room status LEDs
- Display resolution of 0.0001"
- Door status monitor
- Variable alarm delay
- NIST certification included
- Optional BACnet MSTP
- CE and RoHS compliant
- Password enabled
- Positive and negative pressure monitoring
Description
The Setra SRPM Room Pressure Monitor is designed for critical low differential pressure applications that require stringent monitoring and alarming. The SRPM can be configured to monitor either positive-pressure clean rooms such as pharmaceutical processing areas as well as negative-pressure environments such as hospital isolation rooms per CDC guidelines. The SRPM is available with optional BACnet network capability for integration in building automation control systems.
The Setra SRPM Room Pressure Monitor is available in ranges up to ±5.0 inH2O or ±1000 Pa. Standard accuracy is ±0. 50% FS with optional ±0.25% FS accuracy available. The SRPM is a complete system that includes a convenient backlit RGB LCD display with an intuitive graphic user interface for pressure, security, calibration, and alarm setup. The convenient touch-screen allows finger-tip access to menus that guide the user through setup. Visual red and green LEDs and a local audible alarm, with a time delay feature to prevent nuisance alarms, alert personnel to system status. The SRPM also features a SPST relay output for remote indication of system status. Three analog outputs are available in the same unit to interface with BAS system.
Documents
Accessories
Applications
This product can be used in the following applications:
Here's how some of our customers use this product...
Monitoring Clean Rooms in Pharmaceutical Manufacturing
The Background: Our customer is a pharmaceutical company looking for a pressure sensor suitable for monitoring the positive pressure inside a clean room at their facility.
The Problem: The customer needs a solution capable of very accurate measurement of low differential pressures. They also need something immediately.
The Solution: The Setra SRPM Room Pressure Monitor is designed specifically for critical low differential pressure applications that require stringent monitoring and alarming. Setra's reputation within the field, high accuracy/reliability and fast delivery were all taken into consideration prior to purchase.
Monitoring Environmental Air Pressure in a Calibration Lab
The Background: Our customer is a calibration lab that needs to monitor the room pressure to calculate the boiling temperature of liquid helium and liquid nitrogen.
The Problem: The boiling point of a liquid is the temperature at which the vapor pressure of the liquid equals the pressure surrounding the liquid and the liquid changes into a vapor. The boiling point of a liquid varies depending upon the surrounding environmental pressure. Lower environmental pressure results in lower boiling points and higher environmental pressure results in higher boiling points. Liquid helium and liquid nitrogen which both have very low boiling points are especially affected by minute changes in environmental pressure.
The Solution: We recommended the Setra Room Pressure Monitor as it has the accuracy to allow precision measurements while being very simple to use.
Need Help? Call a Calibration engineer at 1-800-884-4967
We're open Mo-Th 8am to 5:30pm. Fr 8am to 5pm ET






















